The Keller and Heckman LLP E-Vapor and Tobacco Law Symposium provided in-depth discussions on the...

5 minute read
The FDA’s Authorisation of Zyn: A Robust PMTA in a Changing Landscape
On January 16, 2025, the FDA granted marketing authorisation for Swedish Match’s Zyn nicotine pouches, marking a significant regulatory milestone for the modern oral nicotine market. Given the rapid expansion of this category, many companies may see...
Filter
Filter
Results - 114 Blogs

Nicotine pouches are exciting products that can help support worldwide tobacco harm reduction....
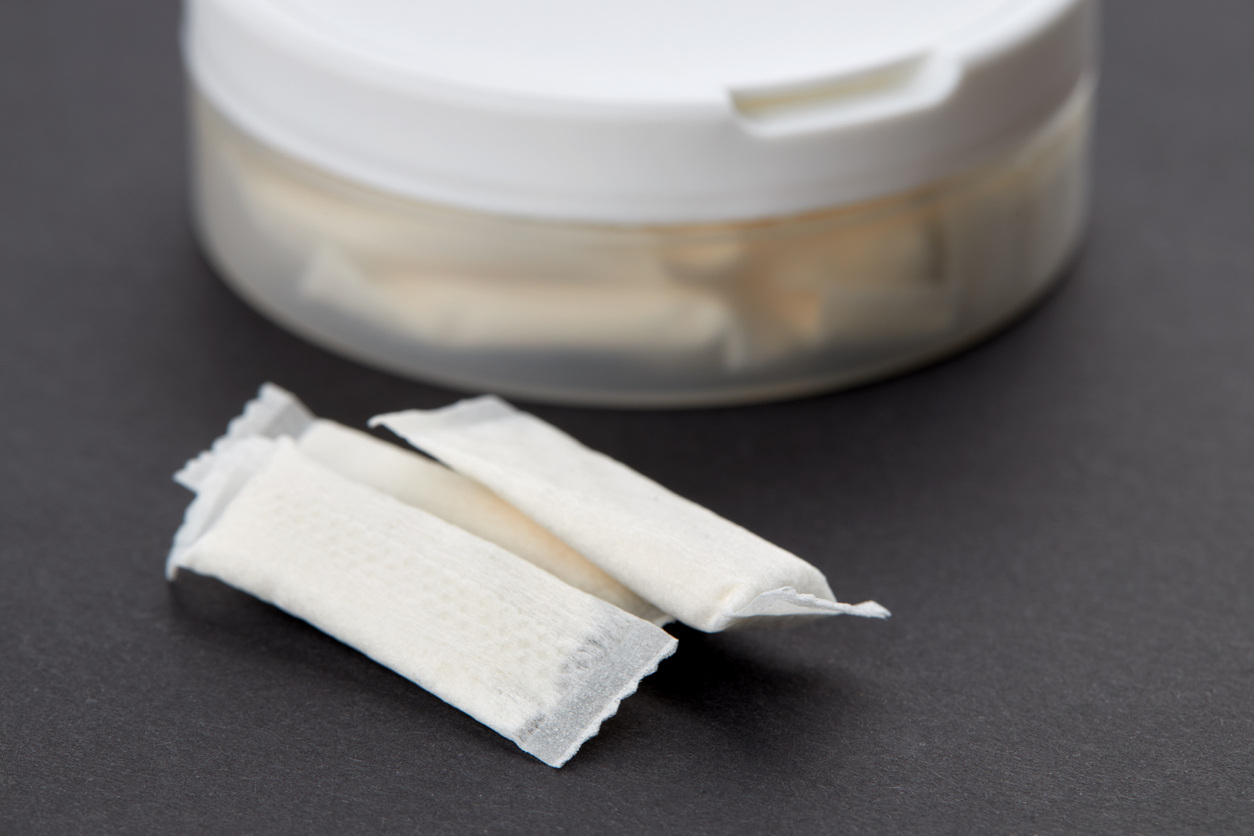
As the global tobacco and nicotine market evolves, nicotine pouches are emerging as a popular...

Broughton welcomes the US Food and Drug Administration’s (FDA) decision to authorize ZYN nicotine...

3 minute read
Navigating FDA and ICH Guidelines: Analytical Testing Validation for Tobacco Products
Navigating FDA and ICH Guidelines: Analytical Testing Validation for Tobacco Products
-2.jpg)
1 minute read
Oral Nicotine Pouches: Trends to Watch in the U.S. and Global Markets during 2025
Oral nicotine pouches have become a disruptive force in the reduced-risk nicotine product...

Allergies concerns highlights need to meet stringent regulations ~

4 minute read
2025 Trends in Tobacco Harm Reduction: Innovation, Regulation, and Market Dynamics
The tobacco harm reduction (THR) industry is undergoing significant changes, driven by advancements...

~ Why new age verification technology could help solve youth appeal ~
Sign up to our monthly bulletins
Ensure your business stays ahead of the competition by keeping up to date on the latest regulatory changes. Sign up for our newsletter to have the latest information and insights served directly to your inbox.
